Intro
Discover the significance of molar weight in chemistry, exploring its impact on molecular structure, chemical reactions, and calculations, with 5 ways to apply molar mass, molecular weight, and atomic mass for accurate results.
The concept of molar weight, also known as molecular weight, is a fundamental aspect of chemistry that plays a crucial role in understanding the properties and behavior of substances. Molar weight is defined as the sum of the atomic weights of the atoms in a molecule, and it is a key factor in determining the physical and chemical properties of a substance. In this article, we will explore five ways that molar weight affects the properties and behavior of substances, and why it is an essential concept in chemistry.
The importance of molar weight cannot be overstated, as it has a significant impact on the physical and chemical properties of substances. From the density and boiling point of a substance to its reactivity and solubility, molar weight plays a crucial role in determining how a substance behaves under different conditions. Furthermore, understanding molar weight is essential for chemists and researchers who need to work with substances in a laboratory setting, as it allows them to predict and control the behavior of substances during experiments and reactions.
In addition to its practical applications, the concept of molar weight is also fascinating from a theoretical perspective. The fact that the properties of a substance can be predicted and understood based on the sum of the atomic weights of its atoms is a testament to the power and simplicity of the laws of chemistry. By exploring the ways in which molar weight affects the properties and behavior of substances, we can gain a deeper understanding of the underlying principles of chemistry and develop new insights into the nature of matter itself.
Understanding Molar Weight
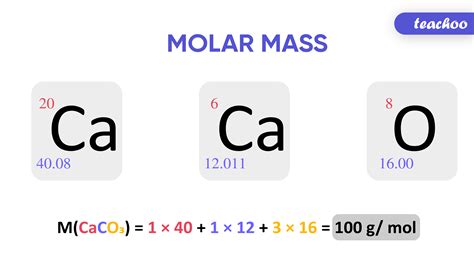
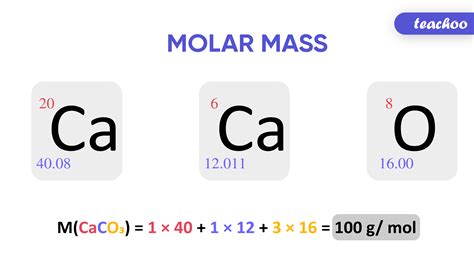
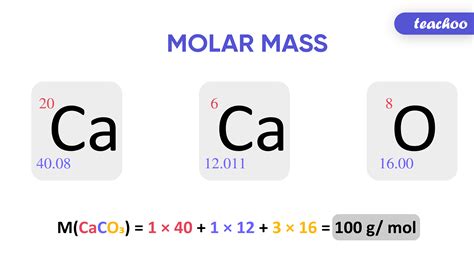
For example, the molar weight of water (H2O) can be calculated by summing the atomic weights of its constituent atoms: hydrogen (H) has an atomic weight of 1.01 g/mol, and oxygen (O) has an atomic weight of 16.00 g/mol. Therefore, the molar weight of water is 2(1.01 g/mol) + 16.00 g/mol = 18.02 g/mol.
Calculating Molar Weight
The calculation of molar weight is a straightforward process that involves summing the atomic weights of the atoms in a molecule. The following steps can be used to calculate the molar weight of a substance: * Determine the chemical formula of the substance * Look up the atomic weights of the atoms in the substance * Sum the atomic weights of the atoms to determine the molar weightFor example, the chemical formula for glucose (C6H12O6) can be used to calculate its molar weight as follows:
- Carbon (C) has an atomic weight of 12.01 g/mol
- Hydrogen (H) has an atomic weight of 1.01 g/mol
- Oxygen (O) has an atomic weight of 16.00 g/mol
- The molar weight of glucose is 6(12.01 g/mol) + 12(1.01 g/mol) + 6(16.00 g/mol) = 180.16 g/mol
Effects of Molar Weight on Physical Properties

For example, the density of a substance is directly related to its molar weight, with substances having higher molar weights tend to have higher densities. This is because the mass of a substance is directly proportional to its molar weight, and the density of a substance is defined as its mass per unit volume.
Similarly, the boiling point and melting point of a substance are also related to its molar weight. Substances with higher molar weights tend to have higher boiling points and melting points than substances with lower molar weights, due to the increased strength of the intermolecular forces between the molecules.
Relationship Between Molar Weight and Boiling Point
The relationship between molar weight and boiling point is a complex one, and it is influenced by a number of factors, including the strength of the intermolecular forces between the molecules and the shape and size of the molecules.In general, substances with higher molar weights tend to have higher boiling points than substances with lower molar weights, due to the increased strength of the intermolecular forces between the molecules. However, there are many exceptions to this rule, and the boiling point of a substance can be influenced by a number of other factors, including its molecular shape and size.
For example, the boiling point of water (H2O) is 100°C, while the boiling point of ethanol (C2H5OH) is 78.5°C. Despite the fact that ethanol has a higher molar weight than water, its boiling point is lower due to the weaker intermolecular forces between its molecules.
Effects of Molar Weight on Chemical Properties

For example, the reactivity of a substance is directly related to its molar weight, with substances having higher molar weights tend to be less reactive than substances with lower molar weights. This is because the larger and more complex molecules of substances with higher molar weights are less able to participate in chemical reactions, due to the increased energy required to break and form chemical bonds.
Similarly, the solubility of a substance is also related to its molar weight, with substances having higher molar weights tend to be less soluble than substances with lower molar weights. This is because the larger and more complex molecules of substances with higher molar weights are less able to interact with the molecules of a solvent, resulting in lower solubility.
Relationship Between Molar Weight and Solubility
The relationship between molar weight and solubility is a complex one, and it is influenced by a number of factors, including the strength of the intermolecular forces between the molecules and the shape and size of the molecules.In general, substances with higher molar weights tend to be less soluble than substances with lower molar weights, due to the decreased ability of the molecules to interact with the molecules of a solvent. However, there are many exceptions to this rule, and the solubility of a substance can be influenced by a number of other factors, including its molecular shape and size.
For example, the solubility of sugar (C6H12O6) in water is high, despite its relatively high molar weight. This is because the molecules of sugar are able to interact with the molecules of water through hydrogen bonding, resulting in high solubility.
Applications of Molar Weight

For example, the molar weight of a substance can be used to determine its density, boiling point, and melting point, as well as its reactivity and solubility. This information can be used to predict the behavior of a substance under different conditions, and to design and optimize chemical reactions and processes.
Additionally, the molar weight of a substance can be used to calculate the amount of the substance required for a reaction, and to predict the outcome of the reaction. This information is essential for chemists and researchers who need to work with substances in a laboratory setting, as it allows them to predict and control the behavior of substances during experiments and reactions.
Importance of Molar Weight in Laboratory Settings
The importance of molar weight in laboratory settings cannot be overstated, as it allows chemists and researchers to predict and control the behavior of substances during experiments and reactions.For example, the molar weight of a substance can be used to calculate the amount of the substance required for a reaction, and to predict the outcome of the reaction. This information is essential for chemists and researchers who need to work with substances in a laboratory setting, as it allows them to predict and control the behavior of substances during experiments and reactions.
Additionally, the molar weight of a substance can be used to determine its properties and behavior, including its density, boiling point, and melting point, as well as its reactivity and solubility. This information can be used to predict the behavior of a substance under different conditions, and to design and optimize chemical reactions and processes.
Molar Weight Image Gallery




What is molar weight and why is it important?
+Molar weight is a measure of the total mass of a molecule, and it is calculated by summing the atomic weights of the atoms that make up the molecule. Molar weight is important because it allows us to predict and control the behavior of substances during experiments and reactions.
How is molar weight calculated?
+Molar weight is calculated by summing the atomic weights of the atoms that make up a molecule. The atomic weight of an atom is a measure of its mass relative to the mass of a standard atom, such as carbon-12.
What are some of the effects of molar weight on physical properties?
+Molar weight has a significant impact on the physical properties of a substance, including its density, boiling point, and melting point. Substances with higher molar weights tend to have higher densities, boiling points, and melting points than substances with lower molar weights.
In conclusion, molar weight is a fundamental concept in chemistry that plays a crucial role in understanding the properties and behavior of substances. The five ways that molar weight affects the properties and behavior of substances that we discussed in this article - understanding molar weight, effects of molar weight on physical properties, effects of molar weight on chemical properties, applications of molar weight, and importance of molar weight in laboratory settings - demonstrate the significance of this concept in chemistry and other fields. We hope that this article has provided you with a deeper understanding of molar weight and its importance, and that you will continue to explore and learn more about this fascinating topic. If you have any questions or comments, please don't hesitate to reach out to us. We would love to hear from you and help you in any way that we can.
